FREQUENCY
OF EXTENDED SPECTRUM BETA LACTAMASE PRODUCING GRAM NEGATIVE BACILLI AMONG
CLINICAL ISOLATES AT CLINICAL LABORATORIES OF ARMY MEDICAL COLLEGE, RAWALPINDI
Ali AM, Rafi S, Qureshi AH
Microbiology Department, Army Medical College Rawalpindi, Pakistan
Background: This study was carried out in
Microbiology department of Army Medical College, Rawalpindi to find out the
frequency of extended spectrum beta lactamase producing gram negative bacilli
among clinical isolates recovered from clinical specimens received from Military Hospital, Rawalpindi. Methods: This study was carried out
from 1st Jan 2002 to 30th Dec 2002. A total of 812 consecutive
Gram-negative bacilli were recovered during the study period from various
samples including urine, blood, pus, sputum, high vaginal swab (HVS),
aspirates, i/v canula/ Central venous lines (CVP), chest tubes and catheter
tips. Extended spectrum beta lactamase detection in these isolates was carried
out by Kirby Bauer disc diffusion method on Mueller Hinton agar. A
susceptibility disk containing amoxicillin-clavulanate was placed as the inhibitor
of beta lactamase in the center of the plate, and cefotaxime,
ceftazidime, ceftriaxone and aztreonam disks were placed 30 mm (center to
center) from the amoxicillin-clavulanate disk. Enhancement of the
zone of inhibition of the oxyimino-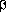 -lactam caused
by the synergy of the clavulanate in the amoxicillin-clavulanate
disk was considered as evidence of ESBL production. Escherichia coli ATCC 25922 and K. pneumoniae
ATCC 700603 were used as control strains. Results:
The frequency of ESBL producing gram negative bacilli among the clinical
isolates was 45%.
-lactam caused
by the synergy of the clavulanate in the amoxicillin-clavulanate
disk was considered as evidence of ESBL production. Escherichia coli ATCC 25922 and K. pneumoniae
ATCC 700603 were used as control strains. Results:
The frequency of ESBL producing gram negative bacilli among the clinical
isolates was 45%.
Keywords: ESBL. Antibiotic.
Nosocomial. Enterobacteriaceae
INTRODUCTION
ß-Lactam
antibiotics are among the most frequently prescribed antimicrobial
agents worldwide. The emergence of resistance to these agents in the
past two decades has resulted in a major clinical crisis.1,2 Gram negative bacteria resistant
to agents such as extended-spectrum cephalosporins, monobactams,
carbapenems and ß-lactam–ß-lactamase inhibitor combinations
have emerged through the production of a variety of ß-lactamases,
alterations in the penicillin-binding proteins, outer membrane
permeability and combinations of multiple mechanisms of resistance.
This increase has paralleled the introduction, administration and
overuse of ß-lactam drugs.3
Resistance
to ß-lactam antimicrobial agents, especially extended-spectrum
cephalo-sporins and other antimicrobial agents among clinical
isolates of gram negative bacteria is on the rise worldwide.4,5
These antimicrobial resistant pathogens include extended-spectrum
cephalosporin resistant Escherichia
coli, Klebsiella pneumoniae,
Enterobacter cloacae, Serratia marcescens and Citrobacter freundii, Pseudomonas aeruginosa and Acinetobacter baumannii.
Recent studies in Taiwan have demonstrated
a high prevalence of these antimicrobial-resistant bacteria and a
trend of increasing resistance under continued antibiotic selective
pressure.6
The
objective of this study was to determine the frequency of extended-spectrum
ß-lactamase (ESBL) producing gram-negative bacilli recovered from clinical
specimens in our set up.
MATERIAL AND METHODS
A total of 812 consecutive
non duplicate gram-negative bacilli recovered from clinical specimens during
the study period from 1st Jan
2002 to 30th Dec 2002 were included in
the study. These were isolated from various samples including urine, blood, pus
aspirate, sputum, chest tube, HVS, i/v canula/ CVP lines and catheter tips
received from patients admitted in Military Hospital, Rawalpindi. The samples
received were initially inoculated on Blood agar and MacConkey`s agar besides
Chocolate agar (in case of sputum) where appropriate. Urine samples were
cultured on Cystiene lactose electrolyte deficient agar (CLED). The samples
were incubated at 37oC under aerobic conditions for 24 hours. The
organisms were primarily identified by standard techniques. Confirmation to the
species level was done by API 20 E & API 20 NE where required. ESBL
production was detected by placing a susceptibility disk containing
amoxicillin-clavulanate (20/10 ug) as the inhibitor of beta lactamase in
the center of the plate and cefotaxime (30ug), ceftazidime (30ug), ceftriaxone
(30ug) and aztreonam (30ug) disks at 30 mm (center to center) from the
amoxicillin-clavulanate disk. Enhancement of the zone of inhibition
of the oxyimino-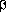 -lactam caused
by the synergy with clavulanate in the amoxicillin-clavulanate disk
was considered as evidence of ESBL production.7 Escherichia coli ATCC 25922 and Klebsiella pneumoniae ATCC 700603 were
used as control strains. The results were tabulated as frequencies.
-lactam caused
by the synergy with clavulanate in the amoxicillin-clavulanate disk
was considered as evidence of ESBL production.7 Escherichia coli ATCC 25922 and Klebsiella pneumoniae ATCC 700603 were
used as control strains. The results were tabulated as frequencies.
RESULTS
Three hundred and sixty six
isolates were ESBL producers making a frequency of 45 %. Enterobacter cloacae
was the most frequent ESBL producer. Escherichia coli (45%) was the most
frequent organism isolated followed by Klebsiella pneumoniae (21%), Pseudomonas
aeruginosa (19.2%), Enterobacter cloacae (4.6%) and Acinetobacter baumannii
(4.4%) (Table-1).
DISCUSSION
Extended spectrum beta lactamase (ESBL) refers
to beta lactamase enzymes produced by gram negative organisms that confer
resistance against broad-spectrum beta-lactam antibiotics, normally having
activity against gram-negative bacilli. Examples of such antibiotics are
cefotaxime, ceftriaxone, ceftazidime and aztreonam.3
The first hospital
outbreak of an ESBL producing gram-negative organism was reported in Germany in
1983.8 Within one year, nosocomial outbreaks caused by a multidrug
resistant Klebsiella clone
carrying a TEM-3 gene were described in France.9 Over the past
decade, ESBL-producing Enterobacteriaceae
have emerged as serious nosocomial pathogens throughout Europe.10
Outbreaks have occurred among the most critically ill patients in intensive
care units (ICUs).11
The prevalence of
ESBL-producing bacteria in most hospitals remains unknown inspite of numerous
reports of nosocomial outbreaks of infection due to these organisms. Important ESBL producing gram-negative bacilli include Klebsiella pneumoniae, Escherichia
coli, and Proteus mirabilis, enterobacter species, Citrobacter freundii, Pseudomonas aeruginosa, Acinetobacter and Stenotrophomonas maltophilia.12
The percentage of
isolates expressing ESBL production is variable although a recent study from
the United States reported 83
ESBL-producing isolates from 906 consecutive isolates of Enterobacteriaceae
over a 20 week period. Klebsiella pneumoniae and Escherichia coli were the most
frequently associated with ESBL production in this study.12
During a five-year surveillance study in
northern France, the overall proportion of ESBL producers was 11.4% in the 6121
strains of Klebsiella species and 47.7% in the 2353 strains of Enterobacter aerogenes.13
In a national
surveillance program conducted in 1996 in Argentina, resistance to
extended-spectrum cephalosporins was shown in 48%, 26%, and 8% of K. pneumoniae, Proteus mirabilis, and
Escherichia coli isolates
respectively.14
In a study carried
out in All India Institute of Medical Sciences, New Delhi during March to
June 2001 out of the 678 isolates tested 68% were ESBL producers, which
are more than that found in our study.
ESBL production was most common in Klebsiella spp. (80%).15
In our study Enterobacter cloacae (79%) was the
most frequent ESBL producer followed by Acinetobacter
baumannii (72%) and Klebsiella
oxytoca (66.66%).
Table-1:
Frequency of gram negative bacilli and ESBLS among the recovered isolates
(n=812)
|
Organism
|
No. of
isolates
|
% of
organism
|
ESBL
producers
|
% of ESBL
Producers
|
|
E.
coli
|
363
|
45%
|
142
|
39.1%
|
|
K.
pneumoniae
|
171
|
21%
|
97
|
57%
|
|
K.
oxytoca
|
6
|
0.7%
|
4
|
66.66%
|
|
Citrobacter
freundii
|
11
|
1.35%
|
4
|
36.36%
|
|
Enterobacter
cloacae
|
38
|
4.6%
|
30
|
79%
|
|
Acinetobacter
baumanii
|
36
|
4.4%
|
26
|
72%
|
|
Acinetobacter
lwofii
|
8
|
0.98%
|
2
|
25%
|
|
Proteus
mirabilis
|
18
|
2.2%
|
11
|
61%
|
|
Proteus
vulgaris
|
2
|
0.2%
|
1
|
50%
|
|
Aeromonas
hydrophila
|
2
|
0.2%
|
1
|
50%
|
|
Providencia
rettgeri
|
1
|
0.2%
|
0
|
0%
|
|
Pseudomonas
aeruginosa
|
156
|
19.2%
|
47
|
36.36%
|
|
TOTAL
|
812
|
100%
|
366
|
45%
|
However these
bacteria were infrequently recovered. The relative frequency of ESBL producing
Gram negative bacilli in our study is similar to frequency reported from
Islamabad, Pakistan16 with Escherichia coli (48%) being the most
prevalent organism reported as was found in our study.
The frequency of
ESBL producing Gram negative bacilli (35 %) in nosocomial isolates reported
from Armed Forces Institute of Pathology, Rawalpindi is lower than our
study. Klebsiella spp. was the
commonest ESBL producing organism reported followed by Enterobacter cloacae and Escherichia coli whereas Enterobacter
cloacae was the most frequent ESBL producer in our study.17
The
frequency of ESBL producing gram negative bacilli clinical isolates in our
study was 45 %. Enterobacter cloacae (79%) was the
most frequent ESBL producing organism detected followed by Acinetobacter baumanii (72%) and Proteus mirabilis (61%) though Escherichia coli (45%) was commonest
organism identified followed by Klebsiella pneumoniae (21%) and Pseudomonas
aerugenosa beside other gram negative rods
CONCLUSION
The resistance to beta lactam
antimicrobial agents among gram-negative bacilli is on the increase
in our setup. Laboratories can detect ESBL production by simple technique of
Jarlier et al. Bacterial strains resistant to most classes of
antibiotics will continue to emerge unless the inappropriate use of
these drugs is curtailed. Clinicians should consider ESBL production as a
possibility in case of treatment failure with 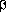 -lactam
antimicrobials.
-lactam
antimicrobials.
REFERENCES
1.
Wood AJ. Antimicrobial-drug resistance. N Eng J Med 1996; 335: 1445–53.
2.
D'Agata FEMC. Antibiotic resistance and exposure
to different generation cephalosporins.
N Eng J Med 2000;28:2678-81.
3.
Fridkin SK, Steward CD,
Edwards JR, Pryor ER, McGowan JE Jr, Archibald LK, et al. Surveillance of
antimicrobial use and antimicrobial resistance in United States hospitals: project
ICARE phase 2. Clin Infect Dis 1999; 29 (2):245–52.
4.
Pfaller MA, Jones RN. MYSTIC (Meropenem Yearly
Susceptibility Test Information Collection) results from the Americas: resistance
implications in the treatment of serious infections. J Antimicrob Chemother
2000; 46(Suppl ):25–37.
5.
Goossens H. MYSTIC (Meropenem Yearly
Susceptibility Test Information Collection) results from Europe: comparison of
antibiotic susceptibilities between countries and centre types. J Antimicrob
Chemother 2000;46 (suppl):39–52.
6.
Hsueh PR, Chen ML, Sun CC, Pan HJ, Yang LS, Ho
SW et al. Emergence of antimicrobial
drug resistance of major pathogens causing nosocomial infections at a
university hospital in Taiwan 1981–1999. Emerg Infect Dis 2002; 8(1):63-8
7.
Jarlier V,
Nicolas M, Fournier G, Philippon A. Extended broad-spectrum 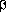 -lactamases
conferring transferable resistance to newer
-lactamases
conferring transferable resistance to newer  -lactam agents
in Enterobacteriaceae: hospital prevalence and susceptibility patterns. Rev
Infect Dis 1988;10 (4):867-78.
-lactam agents
in Enterobacteriaceae: hospital prevalence and susceptibility patterns. Rev
Infect Dis 1988;10 (4):867-78.
8.
Knothe H, Shah P, Krcmery V, Antal M, Mitsuhashi
S. Transferable resistance to cefotaxime, cefoxitin, cefamandole and cefuroxime
in clinical isolates of Klebsiella pneumoniae and Serratia marcescens.
Infection 1983;11(6):315-7.
9.
Brun-Buisson C, Legrand P, Philippon A,
Montravers F, Ansquer M, Duval J et al. Transferable enzymatic resistance to
third-generation cephalosporins during a nosocomial outbreak of multiresistant
Klebsiella pneumoniae. Lancet 1987; 8(2): 302-6.
10.
Livermore DM, Yuan M. Antibiotic resistance and
production of extended spectrum b-lactamases amongst Klebsiella spp. from
intensive care units in Europe. J Antimicrob
Chemother 1996; 38(3):409-24.
11.
Jacoby GA. Extended-spectrum
b-lactamases and other enzymes providing resistance to oxyimino--lactams.
Infect Dis Clin North Am 1997; 11(4):875-87.
12.
Coudron PE, Moland ES, Sanders
CC. Occurrence and Detection of Extended-Spectrum ß-lactamases in members of
the family Enterobacteriaceae at
a Veterans Medical Center: Seek and You May
Find. J Clin Microbiol 1997; 35(10):2593-7.
13.
Albertini MT. Surveillance of
methicillin-resistant Staphylococcus aureus (MRSA) and Enterobacteriaceae
producing extended-spectrum beta-lactamase (ESBLE) in Northern France: a five-year
multicentre incidence study. J Hosp Infect 2002; 52(2): 107-13.
14.
Bantar C, Famiglietti A, Goldberg M. Three-year
surveillance study of nosocomial bacterial resistance in Argentina. The Antimicrobial
Committee; and the National Surveillance Program (SIR) Participants Group. Int
J Infect Dis 2000; 4(2):85-90.
15.
Mathur P, Kapil A, Das B, Dhawan B. Prevalence
of extended spectrum beta lactamase producing gram negative bacteria in a
tertiary care hospital. Indian J Med Res 2002; 115:153-7
16.
Shah AA, Hasan F, Ahmed S, Hameed A.
Extended-spectrum beta-lactamases in enterobact-eriaceae: related to age and
gender. New Microbiol 2002; 25(3): 363-6.
17.
Zaman G, Karamat KA, Abbasi S, Rafi S, Ikram A.
Prevalence of Extended-spectrum beta-lactamase (ESBL) producing
enterobacteriaceae in nosocomial isolates. PAFMJ 1999: 49 (2): 91-6

Address for Correspondence:
Dr. Arif Maqsood Ali, House No. B- 889, Satellite Town, Rawalpindi, Ph :+92-51-4413383,4429514,4842055
Email: drarifmaqsood@hotmail.com
![]() -lactam caused
by the synergy of the clavulanate in the amoxicillin-clavulanate
disk was considered as evidence of ESBL production. Escherichia coli ATCC 25922 and K. pneumoniae
ATCC 700603 were used as control strains. Results:
The frequency of ESBL producing gram negative bacilli among the clinical
isolates was 45%.
-lactam caused
by the synergy of the clavulanate in the amoxicillin-clavulanate
disk was considered as evidence of ESBL production. Escherichia coli ATCC 25922 and K. pneumoniae
ATCC 700603 were used as control strains. Results:
The frequency of ESBL producing gram negative bacilli among the clinical
isolates was 45%.