INTRODUCTION
Papillon-Lefèvre syndrome (PLS)
is a rare autosomal recessive disorder characterized
by hyperkeratosis of the palms and soles and severe destructive periodontal
disease affecting both the primary and permanent teeth. This
case report describes a case of PLS classic clinical features and brief review the relevant literature.
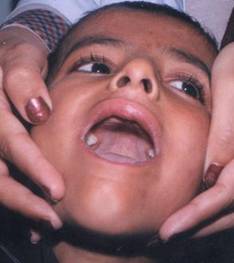
Figure-1:
Absence of upper and lower incisor teeth.
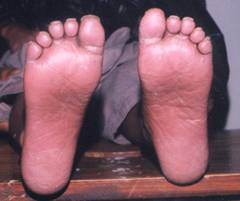
Figure-2: Yellow
colored hyperkeratotic areas on soles
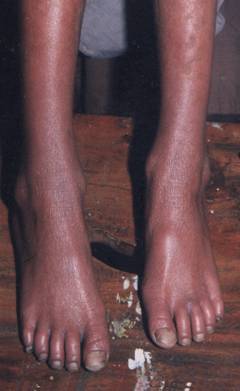
Figure 3
Figure-4
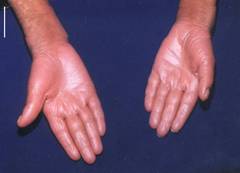
Figure
5
A six year old male child born of cousingeous parents presented to us with redness and
peeling off of skin of hands and feet, cough, redness and discharge from eyes
and ear, pain for last 20 days. He had exacerbations and remissions of the skin
lesions and multiple infections since early childhood. He had normal outcome at
birth and had normal developmental milestones till age of three. During 3rd
year of his life he started developing fissures in skin of palms and soles that
resulted in peeling off of skin leaving red thin skin underneath. He repeatedly
contracted systemic infections. He had cough for last one month that was dry
initially and later on became productive. He used to get bouts of cough and
diagnosed as having pertussis. He complained of
earache that was diagnosed as otitis media. Similarly
he had bilateral purulent conjunctivitis. On examination of oral cavity his
upper incisors, upper lower canines, upper and lower molars were absent (Figure
1). His deciduous teeth started falling off at age of three years and only his
lower incisors erupted again that too became loose. Skin of both palms and
soles was peeling off and underlying skin was red and shiny suggestive of keratoderma (figures 2,3,4,5). The
fingers were pointed and gave a clawed appearance. On eye examination he had
bilateral congested conjunctivae with purulent discharge. Ear examination
revealed bulging red tympanic membrane suggestive of bilateral acute otitis media. On chest auscultation he had bilateral rhonchi. Rest of the systemic examination was normal. On
family history there were no similar disorders in family.
Routine hematological examination revealed Hb% of 10.0 g/dl, total leukocyte count of 9200 and ESR was
20 mm/hour. Urine routine examination showed 15-20 pus cells and 3-4 red blood
cells suggestive of urinary tract infection. Rest of the biochemical
investigations were within normal limits.
Discussion
Papillon-Lefèvre syndrome, or keratosis palmoplantaris with periodontopathia (PLS, MIM 245000), is inherited as an autosomal
recessive trait, affecting children between the ages 1-4 years.1,2
It has a prevalence of 1-4 cases per million persons.2 Males and
females are equally affected and there is no racial predominance.3 PLS
is characterized by palmoplantar keratodermas,
psoriasiform plaques of the elbows and knees,
periodontal disease with resultant premature loss of deciduous and permanent
teeth, and intracranial calcifications.4 These keratotic
plaques may occur focally, but more often involve the entire surface of the
palms and soles.5 Palmoplantar keratosis, varying from mild psoriasiform
scaly skin to overt hyperkeratosis, typically develops within the first three
years of life.6 Often, they are associated hyperhidrosis of the palms and soles resulting in a
foul-smelling odor.2 The findings may worsen in winter and be associated
with painful fissures.7 The development and eruption of the
deciduous teeth proceeds normally, but their eruption is associated with
gingival inflammation and subsequent rapid destruction of the periodontium. The resulting periodontitis
characteristically is unresponsive to traditional periodontal treatment
modalities and the primary dentition is usually exfoliated prematurely by age 4
years. After exfoliation, the inflammation subsides and the gingiva
appears healthy. However, with the eruption of the permanent dentition the
process of gingivitis and periodontitis is usually
repeated and there is subsequent premature exfoliation of the permanent teeth,
although the third molars are sometimes spared.8,9 The etiology of
periodontal disease was explained with juvenile periodontitis
due to infection with Actinobacillus actinomycetemcomitans rather than immunologic dysfunction
or anatomic defects.5 Both
the deciduous and permanent dentitions are affected; resulting in premature
tooth loss. Most PLS patients display both periodontitis
and hyperke-ratosis. Some patients have only palmoplantar keratosis or periodontitis, and in rare individuals the periodontitis is mild and of late onset.6
In addition to the skin and oral findings, patients may have decreased neutrophil, lymphocyte or monocyte
functions and an increased susceptibility to bacteria, associated with
recurrent pyogenic infections of the skin.10
Histologically, the skin lesions of PLS have not been
well-characterized in the literature. Reported findings have consisted of
hyperkeratosis, occasional patches of parakeratosis, acanthosis, and as light perivascular
inflammatory infiltrate4.
The PLS locus has
been mapped to chromosome 11q14–q21 with an almost total loss of cathepsin C activity in PLS patients and reduced activity
in obligate carriers11-13.
The cathepsin C gene encodes a cysteine-lysosomal
protease also known as dipeptidyl-peptidase
I, which functions to remove dipeptides from
the amino terminus of the protein substrate. It also has endopeptidase
activity. The cathepsin-C gene is expressed in
epithelial regions commonly affected by PLS such as palms, soles, knees, and
keratinized oral gingiva. It is also expressed at
high levels in various immune cells including polymorphonuclear
leukocytes, macrophages, and their precursors.14,15
All PLS patients are homozygous for the same cathepsin-C
mutations inherited from a common ancestor. Parents and siblings, heterozygous
for cathepsin C mutations do not show either the palmoplantar hyperkeratosis or severe early onset periodontitis characteristic of PLS8.
A multidisciplinary approach is important
for the care of patients with PLS. The skin manifestations of PPK are usually
treated with emollients7. Oral retinoids
such as acitretin, etretinate,
andisotretinoin were reported to be beneficial for
both dental and cutaneous lesions of PLS. Retinoid
treatment may end up with normal dental development if started during eruption
of permanent teeth.16 The periodontitis in PLS is usually difficult to control.
Effective treatment for the periodontitis includes
extraction of the primary teeth combined with oral antibiotics and professional
teeth cleaning. It is reported that etretinate and acitretin modulate the course of periodontitis
and preserve the teeth. A course of antibiotics should be tried to control the
active periodontitis in an effort to preserve the
teeth and to prevent bacteremia and subsequently pyogenic liver abscess.17
References
1.
Hart TC, Shapira L. Papillon-Lefèvre syndrome. Periodontol 2000 1994;6:88-100.
2.
Gorlin RJ, Sedano H, Anderson VE. The syndrome of
palmar-plantar hyperkeratosis and premature periodontal destruction of the
teeth. J Pediatr 1964;65:895-908.
3.
Cury VF, Costa JE, Gomez RS, Boson WL, Loures CG, De ML. A
novel mutation of the cathepsin C gene in Papillon-Lefèvre syndrome. J Periodontol 2002; 73(3):307-12.
4.
Landow RK, Cheung H, Bauer M. Papillon-Lefèvre syndrome. Int J Dermatol 1983;22:177-9.
5.
Bach JN, Levan NE. Papillon-Lefevre syndrome. Arch Dermatol1968;97:154-8
6.
School of Biological Sciences, University of Manchester, Stopford Building, University of Manchester, Manchester,
UK.
7.
Siragusa M, Romano C, Batticane N, Batolo D, Schepis
C. A new family with Papillon-Lefèvre syndrome: effectiveness of etretinate treatment. Cutis 2000;65(3):151-5.
8.
Hart TC, Hart PS, Bowden DW, Michalec MD, Callison SA, Walker SJ, et al.
Mutations of the cathepsin C gene are responsible for
Papillon-Lefèvre syndrome. J Med Genet 1999;36(12):881-7.
9.
Haneke E. The Papillon-Lefèvre syndrome: keratosis
palmoplantaris with periodontopathy. Report of a case and review of the cases
in the literature. Hum Genet 1979;51(1):1-35.
10. Giansanti JS, Hrabak RP, Waldron CA. Palmar-plantar hyperkeratosis and concomitant periodontal
destruction (papillon-Lefèvre syndrome). Oral Surg Oral Med Oral Pathol 1973;36(1):40-8.
11. Department of Immunology and
Infectious Diseases, New Children's Hospital, Westmead, New South Wales, Australia
12. Department of Pediatric
Dentistry, Westmead Hospital Dental Clinical School, Westmead, New South Wales,
Australia.
13. Department of Oral Diagnosis
and Periodontology, Eins-Shams University, Cairo, Egypt.
14. Angel TA, Hsu S, Kornbleuth
SI, Kornbleuth J, Kramer EM. Papillon-Lefèvre syndrome: a case report of four
affected siblings. J Am Acad Dermatol 2002;46(2 Suppl Case Reports):S8-10.
15. Toomes C, James J, Wood AJ, Wu CL, McCormick D,
Lench N, et al. Loss-of-function mutations in the cathepsin C gene result in
periodontal disease and palmoplantar keratosis. Nat Genet 1999;23(4):421-4.
16. Eickholz P, Kugel B, Pohl S, Naher H, Staehle HJ.
Combined mechanical and antibiotic periodontal therapy in a case of Papillon-Lefevre syndrome. J Periodontol 2001;72:542-9.
17. Almuneef M, Al Khenaizan S, Al Ajaji S, Al-Anazi A.
Pyogenic liver abscess and Papillon-Lefèvre syndrome: not a rare association.
Pediatrics 2003;111(1):e85-8.